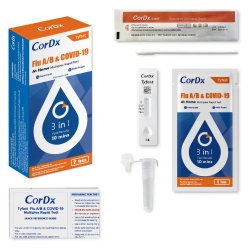
Respiratory Test Kit Tyfast Flu A/B & COVID-19 At Home Multiplex Rapid Test 1 Test per Kit
Features
- CorDx Tyfast Flu A/B & COVID-19 At Home Multiplex Rapid Test is for use under an FDA Emergency Use Authorization (EUA) only: https://www.fda.gov/medical-devices/covid-19-emergency-use-authorizations-medical-devices/in-vitro-diagnostics-euas-antigen-diagnostic-tests-sars-cov-2
- The CorDx Tyfast Flu A/B & COVID-19 At Home Multiplex Rapid Test is a lateral flow immunoassay intended for the qualitative detection and differentiation of SARS-CoV-2, influenza A, and influenza B protein antigens
- This test is authorized for non-prescription home use with self-collected anterior nasal swab specimens from individuals aged 14 years or older, or with adult-collected anterior nasal swab specimens from individuals two (2) years or older
Product Price
$96.13
| McKesson # | 1257160 |
|---|---|
| Manufacturer # | ACT21002110 |
| Brand | Tyfast |
| Manufacturer | CorDx Inc |
| Country of Origin | United States |
| Application | Respiratory Test Kit |
| Buy American Act (BAA) Compliant | Yes |
| Contents 1 | (1) Test Cassette, (1) Swab, (1) Tube with Sample Processing Solution, (1) Quick Ref Instructions |
| Number of Tests | 1 Test per Kit |
| Product Dating | McKesson Acceptable Dating: we will ship >= 90 days |
| Purchase Program Type | Standard Purchase |
| Reading Type | Visual Read |
| Sample Type | Anterior Nasal Swab Sample |
| Specialty | Immunoassay |
| Test Format | Cassette Format |
| Test Kit Type | Rapid |
| Test Method | Immunochromatographic Membrane Assay |
| Test Name | Flu A/B & COVID-19 At Home Multiplex Rapid Test |
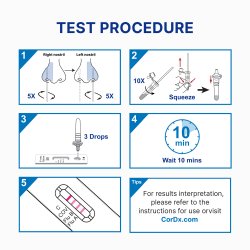
| Time to Results | 10 Minute Results |
| Trade Agreement Act (TAA) Compliant | Yes |
| UNSPSC Code | 41116144 |
Features
- CorDx Tyfast Flu A/B & COVID-19 At Home Multiplex Rapid Test is for use under an FDA Emergency Use Authorization (EUA) only: https://www.fda.gov/medical-devices/covid-19-emergency-use-authorizations-medical-devices/in-vitro-diagnostics-euas-antigen-diagnostic-tests-sars-cov-2
- The CorDx Tyfast Flu A/B & COVID-19 At Home Multiplex Rapid Test is a lateral flow immunoassay intended for the qualitative detection and differentiation of SARS-CoV-2, influenza A, and influenza B protein antigens
- This test is authorized for non-prescription home use with self-collected anterior nasal swab specimens from individuals aged 14 years or older, or with adult-collected anterior nasal swab specimens from individuals two (2) years or older
- This test is only authorized for individuals with signs and symptoms of respiratory infection consistent with COVID-19 within the first five (5) days of symptom onset when tested at least twice over three days with at least 48 hours between tests
- Positive results indicate the presence of viral antigens, but clinical correlation with patient history and other diagnostic information is necessary to determine infection status
- Positive results do not rule out bacterial infection or co-infection with other viruses
- All negative results are presumptive and confirmation with a molecular assay, if necessary for patient management, may be performed
- Negative results do not rule out SARS-CoV-2, influenza A, and influenza B infection and should not be used as the sole basis for treatment or patient management decisions, including infection control decisions such as isolating from others and wearing masks
- Easy to use design, effortless self-administration, whether at home or on the go
- Swift Clarity: Receive results rapidly in 10 minutes for early decision-making
More Information
| Unit of Measure | BX/10 |
|---|
Be the first to review “Respiratory Test Kit Tyfast Flu A/B & COVID-19 At Home Multiplex Rapid Test 1 Test per Kit” Cancel reply
Related products
-
Clinical Laboratories/ Test Kits
Specimen Container McKesson 120 mL (4 oz.) Screw Cap NonSterile
$11.72 – $96.19Price range: $11.72 through $96.19 Select options This product has multiple variants. The options may be chosen on the product page -
Clinical Laboratories/ Test Kits
Specimen Container McKesson 120 mL (4 oz.) Screw Cap Sterile Inside Only
$50.00 Select options This product has multiple variants. The options may be chosen on the product page -
Clinical Laboratories/ Test Kits
McKesson Centrifuge Tube Plain 15 mL Screw Cap Polypropylene Tube
$12.40 Select options This product has multiple variants. The options may be chosen on the product page -
Clinical Laboratories/ Test Kits
Transfer Pipette McKesson 5 mL 0.5 to 1 mL Graduation Increments NonSterile
$50.00 Select options This product has multiple variants. The options may be chosen on the product page






Reviews
There are no reviews yet.